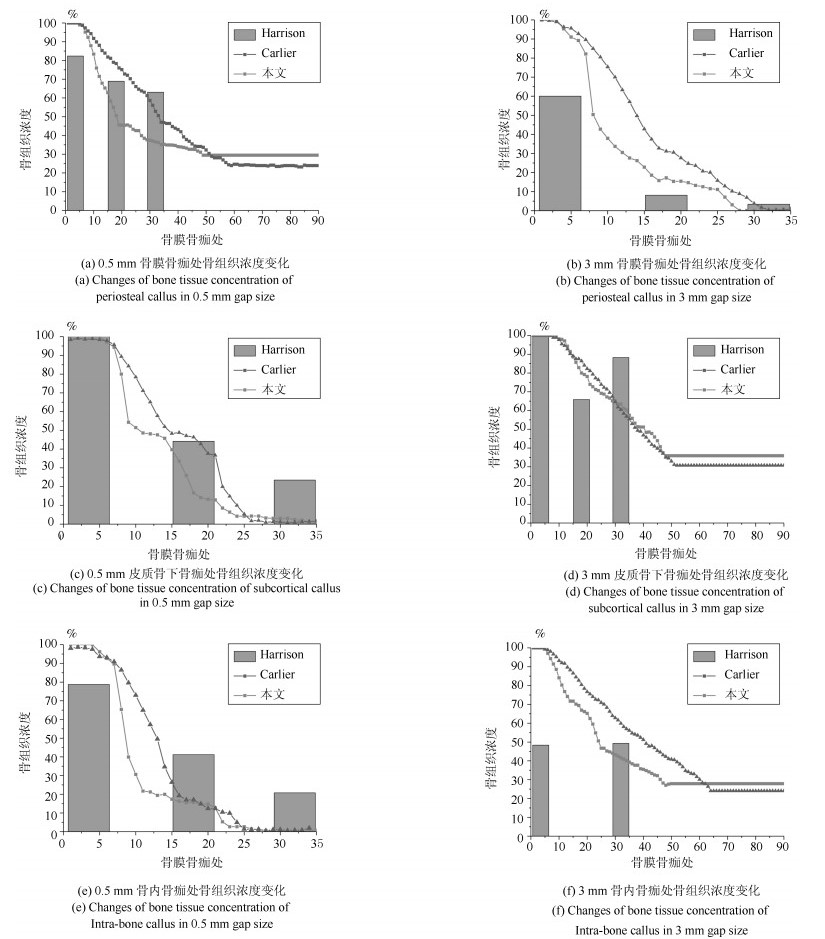
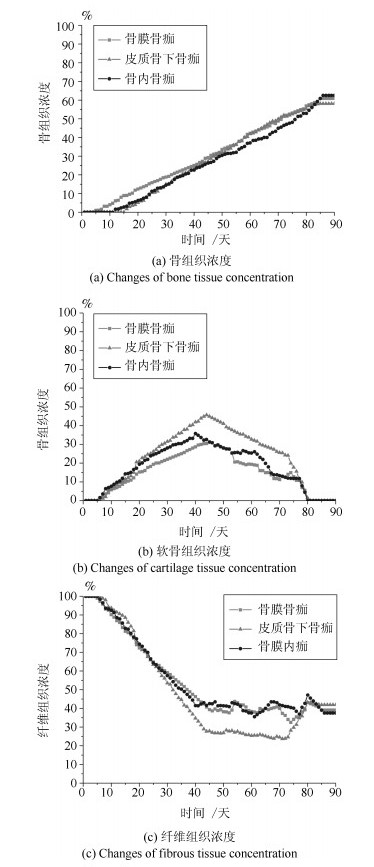
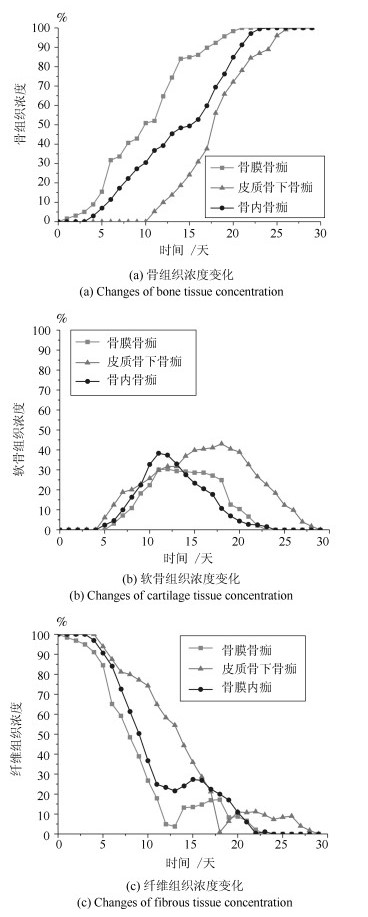
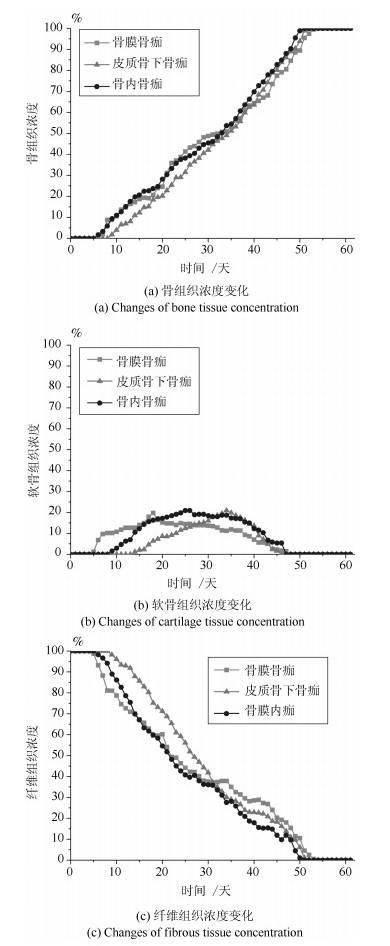
-
摘要: 针对骨折固定的力学条件和组织内的氧气水平, 建立骨折愈合过程的动态仿真模型来描述二期骨折愈合中力学稳定性、组织内氧气水平和组织分化之间的关系.与以往仿真模型不同的是, 本文以三维模型为研究对象, 通过有限元法求解力刺激, 建立偏微分方程求解组织内氧气的时空变化, 采用模糊控制的方法将力和氧气作为主要输入描述组织分化过程, 模拟了骨折愈合过程中的骨折初期、膜内骨化、软骨生成、软骨钙化、骨化等阶段.在Eclipse环境下自主研发骨折愈合过程仿真程序, 通过将本文仿真结果与实验数据、其他模型仿真结果进行对比, 验证了本文的仿真模型和程序的正确性.利用所建立的模型仿真了组织内常氧、低氧、高氧环境下, 两种不同轴向稳定性的鼠横向骨折愈合过程, 得出结论:在低氧或骨间不稳定时, 出现延迟愈合或不愈合; 在常氧环境下, 骨组织正常愈合; 在高氧环境下, 骨折愈合加快.Abstract: In this paper, a simulation model of the dynamic process of fracture healing is established according to the mechanical condition of fracture fixation and oxygen level in tissue to describe the relationship between mechanical stability, intra-tissue oxygen level and tissue differentiation in secondary fracture healing. Unlike previous study models, this paper, a three-dimensional finite element model was established to describe fracture callus mechanics. The spatial and temporal change of oxygen tension in tissues was solved by partial differential equations. The mechanical stimuli and oxygen tension as the main inputs of the fuzzy control to describe the dynamic biological processes of tissue differentiation, including the initial stage of fracture healing, intramembranous ossification, chondrogenesis, cartilage calcification, endochondral ossification and so on. The fracture healing simulation was implemented in the Eclipse. Through the comparison result with experiment data and simulation results in the existing literature, the validity of this model and simulation were verified. We simulate the healing process of transverse osteotomy in rat bone under normoxia, hypoxia and hyperoxia, and analyze the effects of different tissue oxygen levels and different interosseous stability on fracture healing influences. The simulation results show delayed or nonunion of bone healing in hypoxia or interosseous instability, normal healing in normoxia and accelerated fracture healing under hyperoxia.
-
Key words:
- Mechanical status /
- oxygen /
- fracture healing /
- dynamic simulation
1) 本文责任编委 阳春华 -
模型参数 符号 单位 值 氧气扩散系数 $D$ m$^{2}$/s 2.2 $\times$ 10$^{-3}$ 细胞耗氧率 $Q_{{\max}}$ mol/h 9.8 $\times$ 10$^{-14}$ 最大耗氧率
一半时
氧气浓度$K$ mol/mm$^{3}$ 6 $\times$ 10$^{-12}$ 表 2 骨折愈合的模糊规则
Table 2 The fuzzy rules of the fracture healing
6个输入变量 2个输出变量 分化过程 氧张力 骨浓度 邻骨浓度 软骨浓度 膨胀应变 畸变应变 $ \Delta $骨浓度 $ \Delta $软骨浓度 1 – – – – 正过载 – 降低 降低 2 – – – – 负过载 – 降低 降低 3 – – – – – 过载 降低 降低 4 高 – 非低 低 负低 低 增高 – 5 高 – 非低 低 正低 低 增高 – 6 非高 不高 – 非低 负中 非过载 – 增高 7 非高 – – 高 负中 非过载 – 增高 8 非高 – – 非低 负低 非过载 – 增高 9 非低 – 非低 非低 低 零 增高 降低 10 非低 – 非低 非低 负中 零 增高 降低 11 非低 – 非低 非低 负中 低 增高 降低 12 非低 – 非低 非低 负低 低 增高 降低 13 非低 高 – 低 负低 低 增高 降低 14 非低 高 – 低 负低 零 增高 降低 15 非低 – – – 零 零 降低 – -
[1] Pauwels F. A new theory on the influence of mechanical stimuli on the differentiation of supporting tissue the tenth contribution to the functional anatomy and causal morphology of the supporting structure. Zeitschrift fur Anatomie und Entwicklungsgeschichte, 1960, 121: 478-515 doi: 10.1007/BF00523401 [2] Carter D R, Blenman P R, Beaupré G S. Correlations between mechanical stress history and tissue differentiation in initial fracture healing. Clinical Orthopaedics Related Research, 2010, 6(5): 736-748 [3] Blenman P R, Carter D R, Beaupre G S. Role of mechanical loading in the progressive ossification of a fracture callus. Journal of Orthopaedic Research, 1989, 7(3): 398-407 doi: 10.1002/jor.1100070312 [4] Claes L E, Heigele C A, Neidlinger-Wilke C. Effects of mechanical factors on the fracture healing process. Clinical Orthopaedics Related Research, 1998, 355(Suppl.): 132-147 [5] Claes L E, Heigele C A. Magnitudes of local stress and strain along bony surfaces predict the course and type of fracture healing. Journal of Biomechanics, 1999, 31(3): 255-266 [6] Lacroix D, Prendergast P. A mechano-regulation model for tissue differentiation during fracture healing: Analysis of gap size and loading. Journal of Biomechanics, 2017, 35(9): 1163-1171 [7] Isaksson H, Wilson W, van Donkelaar C C. Comparison of biophysical stimuli for mechano-regulation of tissue differentiation during fracture healing. Journal of Biomechanics, 2016, 39(8): 1507-1516 [8] Vetter A, Witt F, Sander O. The spatio-temporal arrangement of different tissues during bone healing as a result of simple mechanobiological rules. Biomechanics and Modeling in Mechanobiology, 2016, 11(2): 147-160 [9] Ament C, Hofer E. A fuzzy logic model of fracture healing. Journal of Biomechanics, 2000, 33: 961-968 doi: 10.1016/S0021-9290(00)00049-X [10] Simon U, Augat P, Claes L. A numerical model of the fracture healing process that describes tissue development and revascularization. Computer Methods in Biomechanics and Biomedical Engineering, 2011, 14(1): 79-93 doi: 10.1080/10255842.2010.499865 [11] Steiner M, Claes L, Ignatius A, et al. Disadvantages of interfragmentary shear on fracture healing-mechanical insights through numerical simulation. Journal of Orthopaedic Research, 2014, 32(7): 865-872 doi: 10.1002/jor.22617 [12] Steiner M, Claes L, Ignatius A. Prediction of fracture healing under axial loading, shear loading and bending is possible using distortional and dilatational strains as determining mechanical stimuli. Journal of the Royal Society Interface, 2013, 10(86): 1123-1126 [13] 王沫楠.基于血液供给条件和力学环境的骨折愈合仿真.自动化学报, 2018, 44(2): 240-250 doi: 10.16383/j.aas.2018.c160673Wang Mo-Nan. Fracture healing simulation considering blood supply and mechanics conditions. Acta Automatica Sinica, 2018, 44(2): 240-250 doi: 10.16383/j.aas.2018.c160673 [14] Burke K. Substrate stiffness and oxygen as regulators of stem cell differentiation during skeletal tissue regeneration: a mechanobiological model. PLoS One, 2012, 7(7): 216-221 [15] Wagegg M, Gaber T, Lohanatha, et al. Hypoxia promotes osteogenesis but suppresses adipo-genesis of human mesenchymal stromal cells in a hypoxia-inducible factor-1dependent manner. PLoS One, 2012, 7(9): e46483 doi: 10.1371/journal.pone.0046483 [16] Meyer E G, Buckley C T, Thorpe S D. Low oxygen tension is a more potent promoter of chondrogenic differentiation than dynamic compression. Journal of Biomechanics, 2010, 43: 2516-2523 doi: 10.1016/j.jbiomech.2010.05.020 [17] Geris L, Gerisch A, Sloten J V. Angiogenesis in bone fracture healing: a bioregulatory model. Journal of Theoretical Biology, 2008, 251(1): 137-158 doi: 10.1016/j.jtbi.2007.11.008 [18] Peiffer V, Gerisch A, Vandepitte D. A hybrid bioregulatory model of angiogenesis during bone fracture healing. Biomechanics and Modeling in Mechanobiology, 2011, 10(3): 383-395 doi: 10.1007/s10237-010-0241-7 [19] Carlier A, Geris L, Bentley K. Mosaic: a multiscale model of osteogenesis and sprouting angiogenesis with lateral inhibition of endothelial cells. Plos Computational Biology, 2012, 8(10): 1371-1387 [20] Burke D, Dishowitz M, Sweetwyne M. The role of oxygen as a regulator of stem cell fate during fracture repair in TSP2-Null mice. Journal of Orthopaedic research, 2013, 31(10): 1585-1596 doi: 10.1002/jor.22396 [21] Harrison L J, Cunningham J L, Stromberg L. Controlled induction of a Pseudarthrosis: A Study Using a Rodent Model. Journal of Orthopaedic Trauma, 2003, 17(1): 11-21 doi: 10.1097/00005131-200301000-00003 [22] Hershey D, Karhan T. Diffusion coefficients for oxygen transport in whole blood. AIChE Journal, 1968, 14(6): 969-972 doi: 10.1002/aic.690140628 [23] Pattappa G, Heywood H K, de Bruijn J D. The metabolism of human mesenchymal stem cells during proliferation and differentiation. Journal of Cellular Physiology, 2011, 226(10): 2562-2570 doi: 10.1002/jcp.22605 [24] Epari D R, Lienau J, Schell H. Pressure, oxygen tension and temperature in the periosteal callus during bone healing——An in vivo study in sheep. Bone, 2008, 43(4): 734-739 doi: 10.1016/j.bone.2008.06.007 [25] Lu C Y, Saless N, Wang X D. The role of oxygen during fracture healing. Bone, 2013, 52(1): 220-229 doi: 10.1016/j.bone.2012.09.037 [26] Murao H, Yamamoto K, Matsuda S. Periosteal cells are a major source of soft callus in bone fracture. Journal of Bone and Mineral Metabolism, 2013, 31(4): 390-398 doi: 10.1007/s00774-013-0429-x [27] Einhorn T A. The cell and molecular biology of fracture healing. Clinical Orthopaedics and Related Research, 1998, 355(Suppl.): S7-S21 [28] Merceron C, Vinatier C, Portron S. Differential effects of hypoxia on osteochondrogenic potential of human adipose derived stem cells. American Journal of Physiology-Cell Physiology, 2010, 298(1): 355-364 [29] 侯增广, 赵新刚, 程龙, 王启宁, 王卫群.康复机器人与智能辅助系统的研究进展.自动化学报, 2016, 42(12): 1765-1779 doi: 10.16383/j.aas.2016.y000006Hou Zeng-Guang, Zhao Xin-Gang, Cheng Long, Wang Qi- Ning, Wang Wei-Qun. Recent advances in rehabilitation robots and intelligent assistance systems. Acta Automatica Sinica, 2016, 42(12): 1765-1779 doi: 10.16383/j.aas.2016.y000006 -





 下载:
下载: